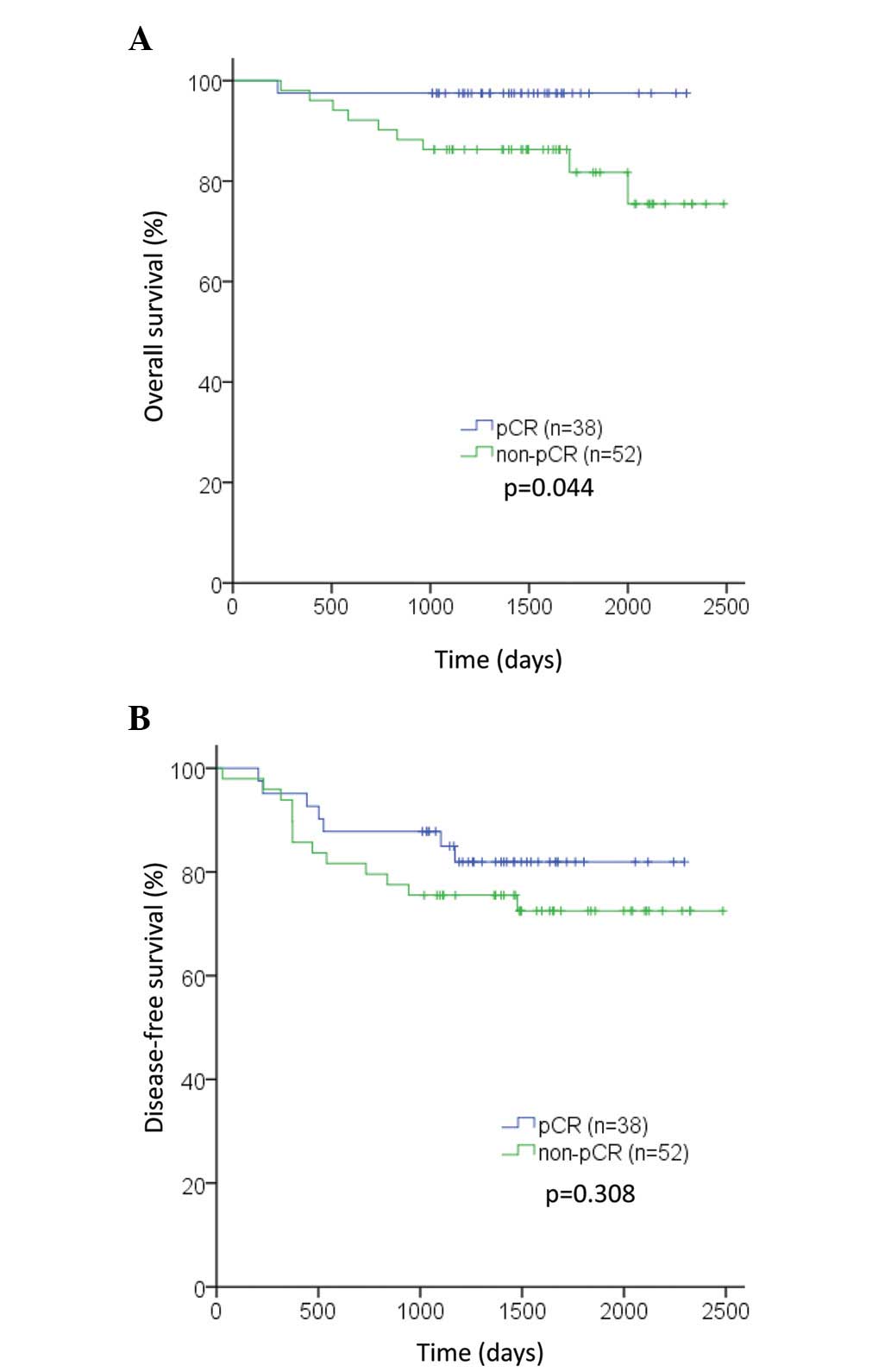
We earlier reported that women with HER2 and ER-positive breast cancer receiving neoadjuvant dual HER2-block and palbociclib in the NA-PHER2 trial had Ki67 decrease and 27% pathological complete responses (pCR). The presence of TILs at diagnosis is an independent, positive, prognostic marker in HER2-positive early breast cancer treated with neoadjuvant anti-HER2 agents and chemotherapy for both pCR and EFS end points.Ĭ Identifier: NCT00553358.The crosstalk between estrogen and HER2 receptors and cell-cycle regulation sustains resistance to endocrine therapy of HER2- and hormone receptor-positive breast cancer. With a median (IQR) follow-up time of 3.77 (3.50-4.22) years, every 1% increase in TILs was associated with a 3% decrease in the rate of an event (adjusted hazard ratio, 0.97 P =. For the pCR end point, levels of TILs greater than 5% were associated with higher pCR rates independent of treatment group (adjusted odds ratio, 2.60 P =. The median (interquartile range ) level of TILs was 12.5% (5.0%-30.0%), with levels lower in hormone receptor-positive (10.0% ) vs hormone receptor-negative (12.5% ) samples (P =. Of the 455 patients, 387 (85.1%) tumor samples were used for the present analysis. Levels of TILs were examined for their associations with efficacy end points adjusted for prognostic clinicopathological factors including PIK3CA genotype. We evaluated levels of percentage of TILs using hematoxylin-eosin-stained core biopsy sections taken at diagnosis (prior to treatment) in a prospectively defined retrospective analysis. The primary end point used in this study was pCR in the breast and lymph nodes, with a secondary end point of EFS. The NeoALTTO trial (Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimization) randomly assigned 455 women with HER2-positive early-stage breast cancer between January 5, 2008, and May 27, 2010, to 1 of 3 neoadjuvant treatment arms: trastuzumab, lapatinib, or the combination for 6 weeks followed by the addition of weekly paclitaxel for 12 weeks, followed by 3 cycles of fluorouracil, epirubicin, and cyclophosphamide after surgery. To determine associations between presence of TILs, pathological complete response (pCR), and event-free survival (EFS) end points in patients with early breast cancer treated with trastuzumab, lapatinib, or the combination. The prognostic associations in the neoadjuvant setting of other anti-HER2 agents and combinations are unknown. The presence of tumor-infiltrating lymphocytes (TILs) is associated with improved outcomes in human epidermal growth factor receptor 2 (HER2)-positive early breast cancer treated with adjuvant trastuzumab and chemotherapy. 12 Breast Cancer Translational Research Laboratory, Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium.11 Centre de Recherche en Epidémiologie et Santé des Populations, INSERM U1018, Service de Biostatistique et d'Epidémiologie, Gustave Roussy, Université Paris-Sud, Villejuif, France.10 Department of Medicine, Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium.9 Memorial Sloan-Kettering Cancer Center, New York, New York.8 GlaxoSmithKline Oncology, Collegeville, Pennsylvania.7 Department of Obstetrics and Gynecology, Campus Kiel, University Hospital Kiel, Kiel, Germany.6 Breast European Adjuvant Study Team, Institut Jules Bordet, Brussels, Belgium.5 Val d'Hebron Institute of Oncology, Barcelona, Spain.4 Division of Clinical Medicine and Research, Peter MacCallum Cancer Centre, University of Melbourne, Melbourne, Victoria, Australia.3 Frontier Science (Scotland) Ltd, Grampian View, Kincraig, Kingussie, United Kingdom.2 Institute of Pathology, Charité-Universitätsmedizin, Berlin, Germany4German Cancer Consortium, Berlin, Germany.1 Breast Cancer Translational Research Laboratory, Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium2Department of Pathology, Gasthuis Zusters Antwerpen Hospitals, Antwerp, Belgium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
